Crossing-first &
Exploring New Frontiers
ProSonic™
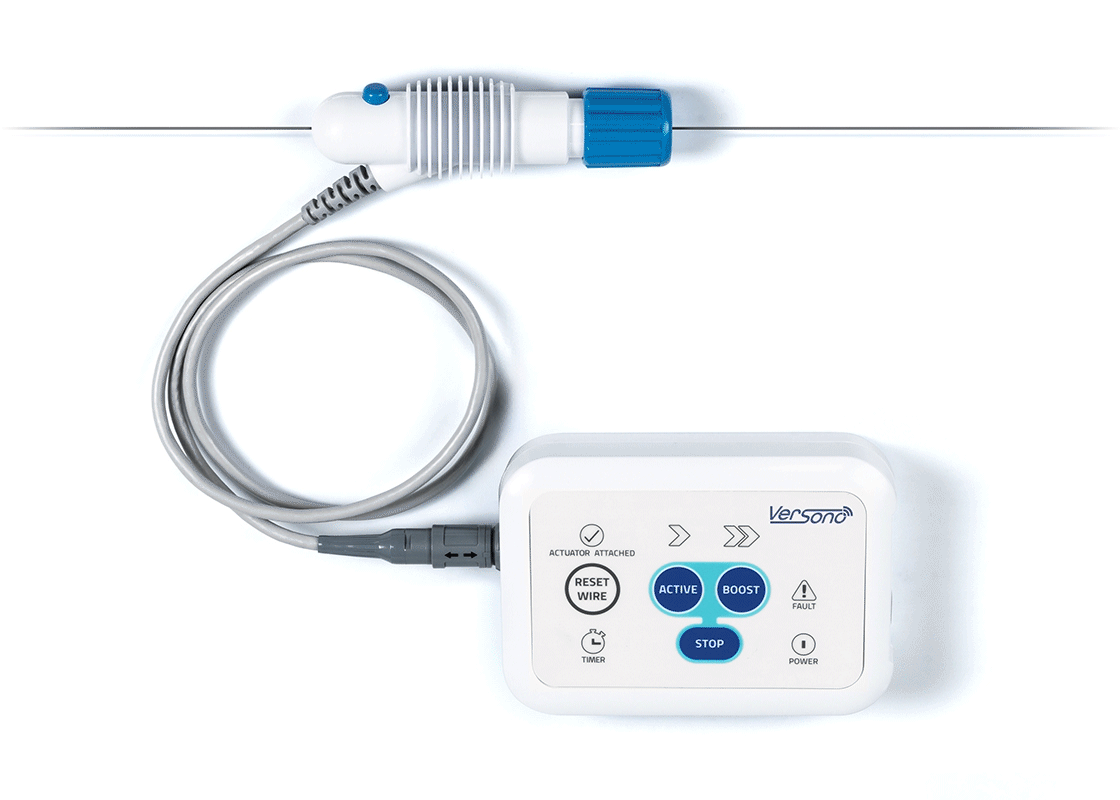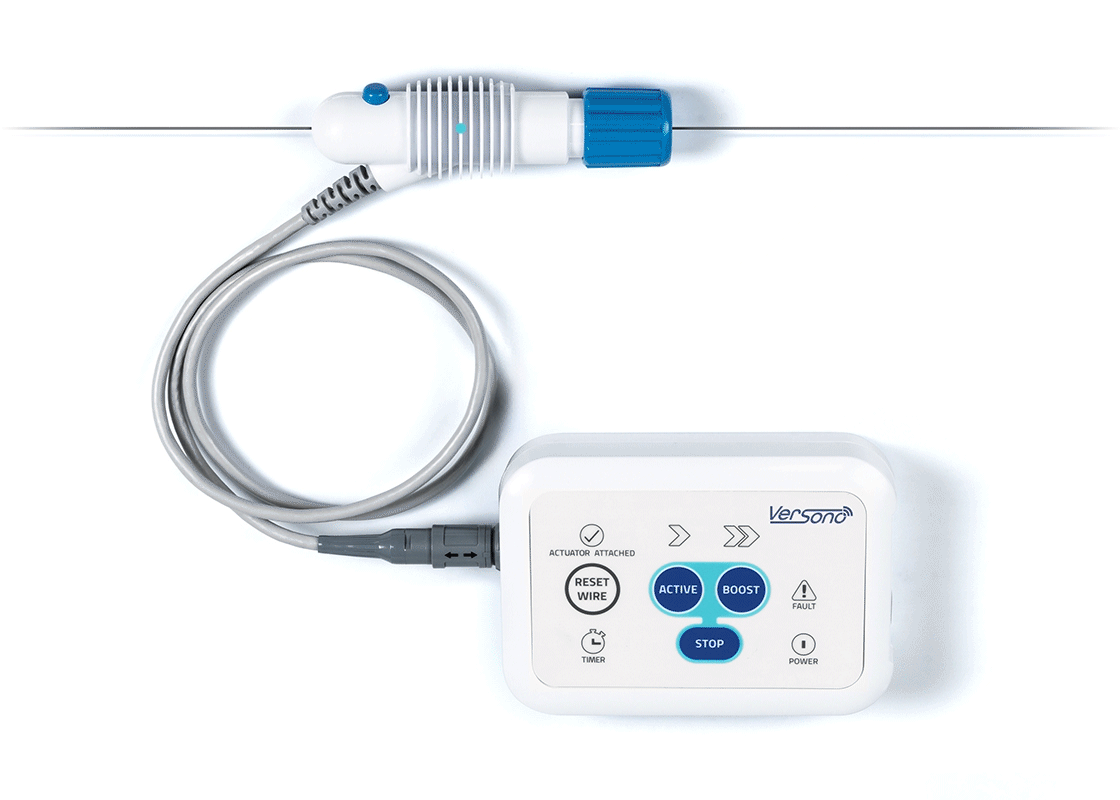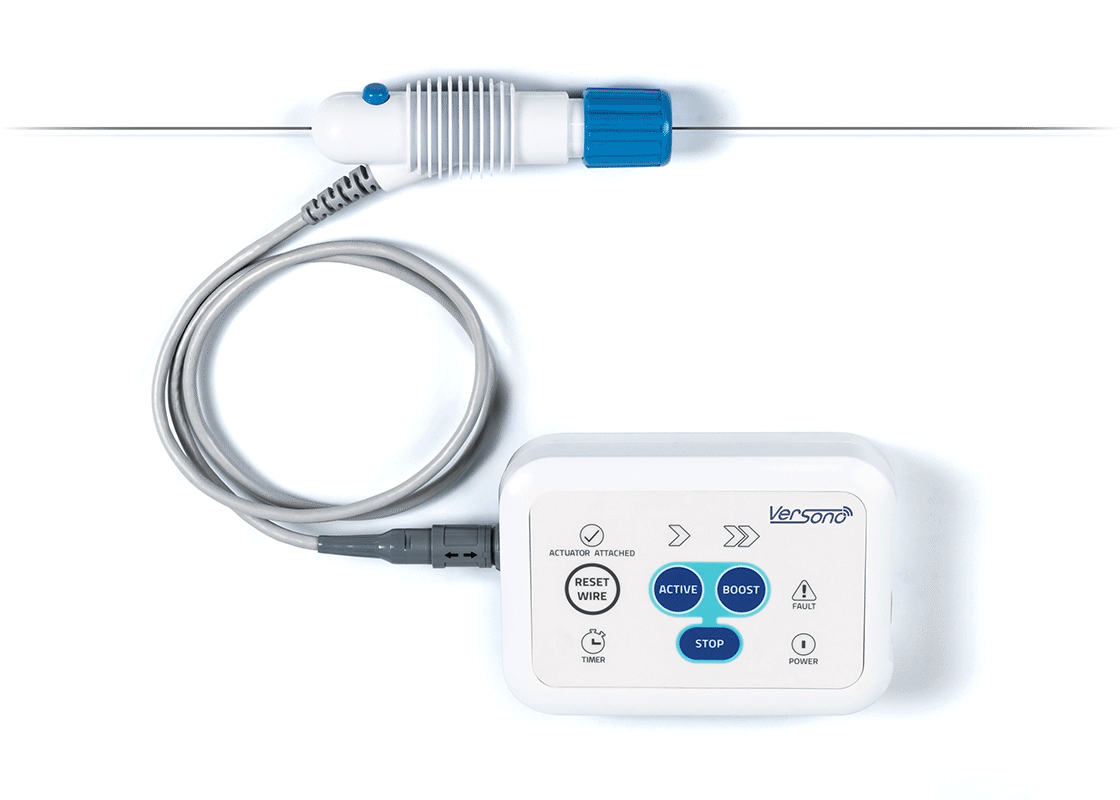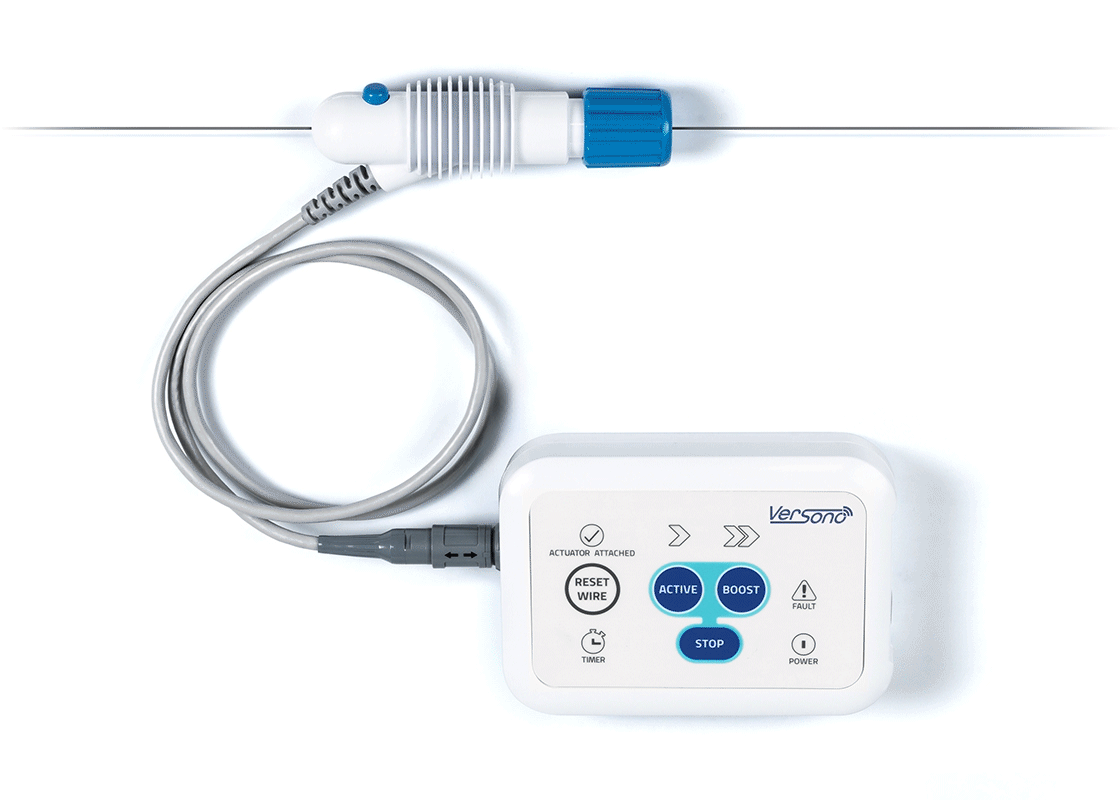
ProSonic™: Built Around the Realities of CTO Crossing
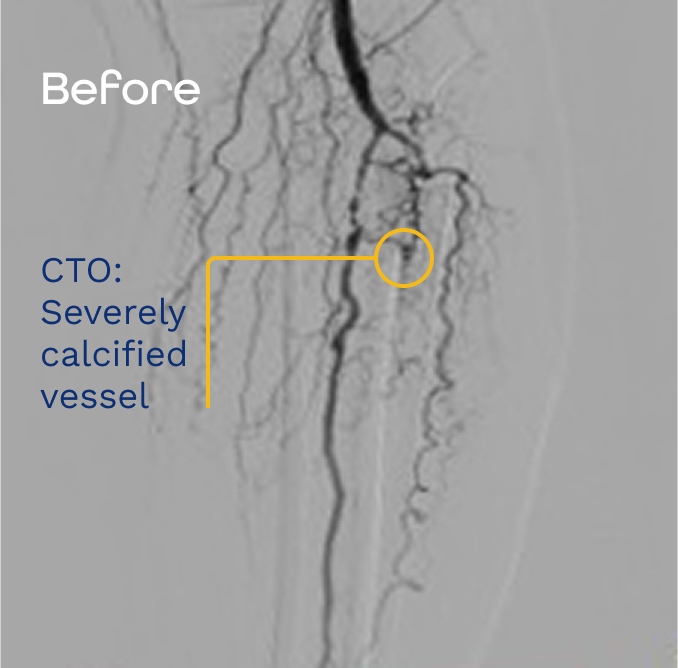
Complex CTOs present challenges that often determine procedural strategy before treatment begins. Dense calcification, ambiguous proximal caps, and limited true lumen access can constrain what follows.
The FastWire® technology is being developed to prioritise the crossing step itself, supporting intraluminal crossing and enabling access for established follow-on therapies, guided by operator judgement.

A Distinct Mechanism of Action Under Evaluation
The device delivers ultrasonic energy intravascularly through a wire-mounted sonotrode.
Energy is delivered:
Axially, along the direction of the vessel during crossing.
Radially, outward from the device into the vessel wall.
These combined axial and radial pressure waves represent a distinct mode of action currently under clinical evaluation.
Ultrasonic Mechanism of Action
The following sequence describes design intent and observed behaviour under evaluation, not clinical outcomes.
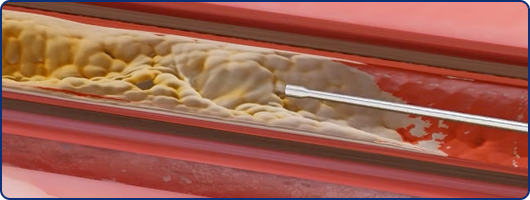
Position
ProSonic™’s tip is positioned adjacent to the calcified occlusive cap. This non-contact positioning provides space for acoustic streaming and cavitation to occur without mechanical energy damping.
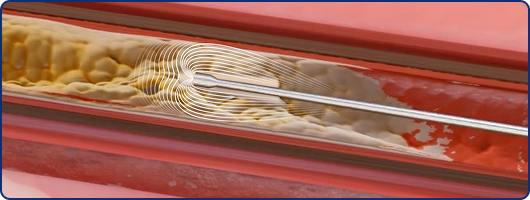
Activation
High-frequency ultrasonic activity, generates high-speed pressure(shock) waves at the tip of the device, with acoustic effects interacting with occlusive disease under clinical evaluation.
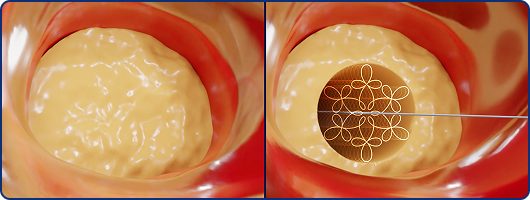
Orbital Modulation
Axial tip movement and cavitation interact as ultrasonic energy propagates, producing a dynamic acoustic pattern along the intended path within the occlusive segment.

Preparation
The combined axial and radial motion establishes a localised ultrasonic energy field along the intended path, shaping the vessel environment ahead of subsequent procedural steps.
Crossing First With More to Follow
Current development and clinical evaluation is focused on enabling crossing.
Future indications, including vessel preparation, are being explored.
Integrates Seamlessly Into Clinical Workflows
The technology is being developed to align with established endovascular approaches, allowing ultrasonic crossing to be incorporated into familiar procedural workflows.
The intent is not to replace existing therapies, but to support access through complex occlusive disease so that standard treatment options remain available.
Clinical Evaluation
ProSonic™ is currently being evaluated in a multi-centre clinical study following first-in-human use, with data contributing to ongoing clinical development and regulatory engagement.
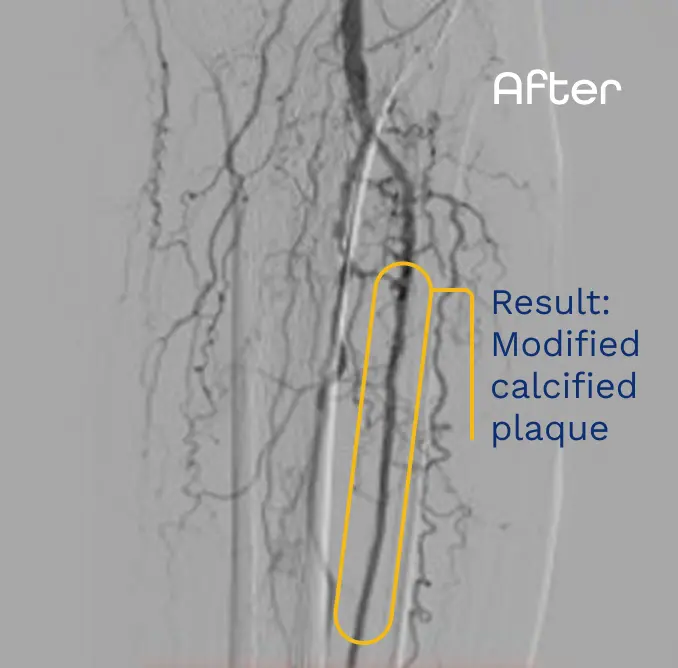
Presented NCVH 2023 by Dr. Craig Walker. Images are for informational purposes only. No claims of clinical benefit are made.
Regulatory Status
ProSonic™ is an investigational device and is not approved or cleared for sale or commercial use in any market. Descriptions reflect development intent and areas of evaluation only and do not represent clinical performance or outcomes.
